
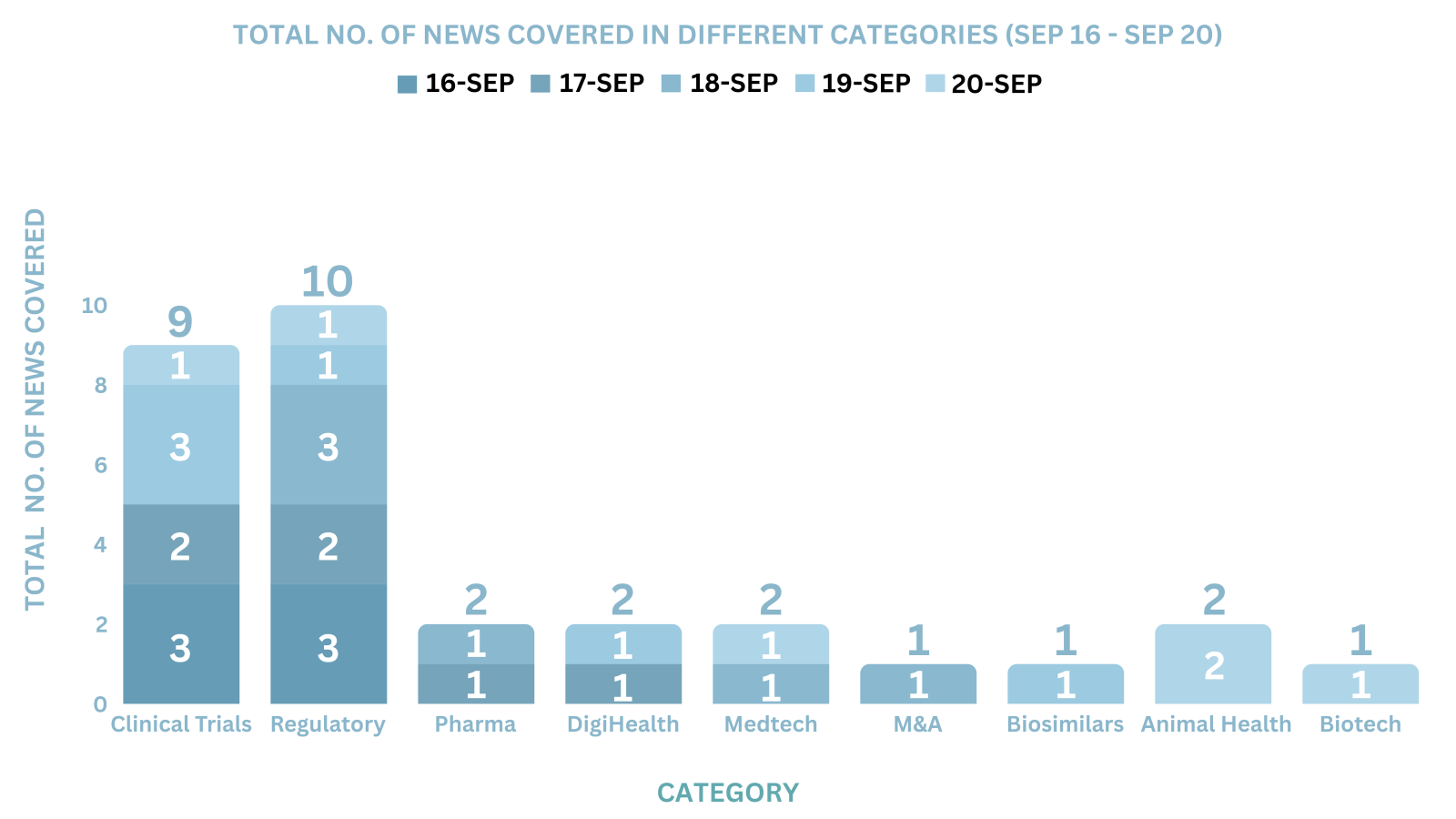
PharmaShots Weekly Snapshots (September 16 – September 20, 2024)
This week PharmaShots’ news was all about the updates on Clinical Trials, Pharma, Biotech, COVID-19, Regulatory & MedTech. Check out our full report below:

Novartis to Highlight Updated Data from the P-III (NATALEE) Study of Kisqali (Ribociclib) for Treating Early Breast Cancer (EBC) at ESMO 2024
Read More: Novartis
Merck Highlights 10 Years Data from the P-III (KEYNOTE-006) Study of Keytruda in Advanced Melanoma Patients at ESMO 2024
Read More: Merck
Boehringer Ingelheim Reports the P-III (FIBRONEER-IPF) Trial Data of Nerandomilast to Treat Idiopathic Pulmonary Fibrosis (IPF)
Read More: Boehringer Ingelheim
Merck and Daiichi Sankyo Report the P-III (HERTHENA-Lung02) Study Data of Patritumab Deruxtecan to Treat Non-Small Cell Lung Cancer
Read More: Merck & Daiichi Sankyo
Johnson & Johnson Reports the P-II (SunRISe-4) Study Data of TAR-200 Plus Cetrelimab to Treat Muscle-Invasive Bladder Cancer
Read More: Johnson & Johnson
Roche Reports Topline Data from P-III (CENTERSTONE) Study of Xofluza to Reduce the Transmission of Influenza Virus
Read More: Roche
GSK Reports Topline Results from the P-III Trial of Arexvy Co-Administered with Shingrix
Read More: GSK
Satellos Reports the First Patient Dosing with SAT-3247 in P-I Trial for Duchenne Muscular Dystrophy
Read More: Satellos
Sanofi Reports the P-III (HERCULES) Study Results of Tolebrutinib for Treating Non-Relapsing Secondary Progressive Multiple Sclerosis (nrSPMS)
Read More: Sanofi

Eli Lilly’s Ebglyss (L ebrikizumab-lbkz) Receives the US FDA’s Approval for Treating Moderate-to-Severe Atopic Dermatitis
Read More: Eli Lilly
Sanofi and Regeneron’s Dupixent Receives the US FDA’s Approval to Treat Adolescents with Chronic Rhinosinusitis with Nasal Polyps (CRSwNP)
Roche Reports the US FDA’s Approval of Ocrevus Zunovo for Treating Relapse and Progressive Multiple Sclerosis
Read More: Roche
GSK Reports the NDA Acceptance of Blenrep Combinations by the Japanese MHLW for R/R Multiple Myeloma
Read More: GSK
Poseida Therapeutics’ P-BCMA-ALLO1 Gains the US FDA’s Regenerative Medicine Advanced Therapy Designation for R/R Multiple Myeloma
Read More: Poseida Therapeutics
AstraZeneca’s Fasenra Receives the US FDA’s Approval for the Treatment of Eosinophilic Granulomatosis with Polyangiitis
Read More: AstraZeneca
Merck Reports the US FDA’s Approval of Keytruda Plus Pemetrexed and Platinum Chemotherapy to Treat Malignant Pleural Mesothelioma (MPM)
Read More: Merck
Glycomine’s GLM101 Gains the US FDA’s Fast Track Designation to Treat Phosphomannomutase 2-Congenital Disorder of Glycosylation (PMM2-CDG)
Read More: Glycomine
Novartis Reports the US FDA’s Approval of Kisqali (Ribociclib) for the Treatment of HR+/HER2- Early Breast Cancer
Read More: Novartis
Johnson & Johnson Reports the US FDA’s Approval of Rybrevant Plus Standard of Care to Treat EGFR-Mutated NSCLC
Read More: Johnson & Johnson

Mindpeak Partners with Roche to Strengthen Cancer Diagnostics Using AI
Read More: Mindpeak & Roche
Astellas Reports the US FDA Listing of DIGITIVA to Manage Heart Failure
Read More: Astellas

Vivtex Partners with Equillium to Develop EQ302 for Gastrointestinal Inflammation
Read More: Vivtex & Equillium
PharmaJet Join Forces with Scancell to Develop and Commercialize Needle-free DNA Vaccine for Advanced Melanoma
Read More: PharmaJet & Scancell

Boston Scientific Reports the US FDA’s Expanded Approval of INGEVITY+ Pacing Leads for Conduction System Pacing
Read More: Boston Scientific
AngioDynamics Commences the RECOVER-AV Study of AlphaVac F1885 System to Treat Pulmonary Embolism
Read More: AngioDynamics

Organon to Acquire Dermavant for an Aggregate of ~$1.2B
Read More: Organon & Dermavant

Accord BioPharma’s Hercessi 420mg (Biosimilar, Herceptin) Receives the US FDA’s Approval to Treat Various HER2-Overexpressing Cancer
Read More: Accord BioPharma

Elanco Animal Health Reports the US FDA’s Approval of Zenrelia (Ilunocitinib) to Treat Canine Dermatological Disorders
Read More: Elanco
epiq Animal Health and ArmOR Hand Gloves Collaborate to Offer Novel Products for Veterinarians within the Animal Health Distribution Channels
Read More: Epiq Animal Health

Rakuten Medical Highlights the Preclinical Results of RM-0256 at JCA 2024
Read More: Rakuten Medical
Related News: PharmaShots Weekly Snapshots (September 09 – September 13, 2024)
Tags

Disha was a content writer at PharmaShots. She is passionate and curious about recent updates and developments in MedTech and Pharma industry. She covers news related to clinical trial results and updates. She can be contacted at connect@pharmashots.com.














